January 19, 2024

Developing CAR T-cell therapies and companion diagnostics requires expertise in discovery, regulatory compliance, biomanufacturing, and long-term real-world evidence generation
Chimeric antigen receptor (CAR) T-cell therapy is a type of advanced immunotherapy for cancer. This treatment has proven successful for some patients with very advanced cancer or in cases where other treatments failed. The personalized nature of CAR T-cell therapy requires the use of companion diagnostics (CDx) — devices that detect specific biomarkers — to determine whether someone is a candidate for the therapy, since it can cause serious adverse events in patients for whom the therapy is not designed.
The dual challenge for manufacturers to effectively demonstrate efficacy for the therapy alongside a CDx is further compounded by a complicated path to regulatory approval and reimbursement. After approval, there is also the additional challenge of biomanufacturing CAR T cells at scale. Navigating this labyrinth to improve patient access and outcomes requires knowledge, expertise, and experience with each critical element of the drug and diagnostic device lifecycle: discovery, development, regulatory compliance, market access, long-term real-world evidence generation, and biomanufacturing.
CAR T-cell therapy: using a patient's own immune cells
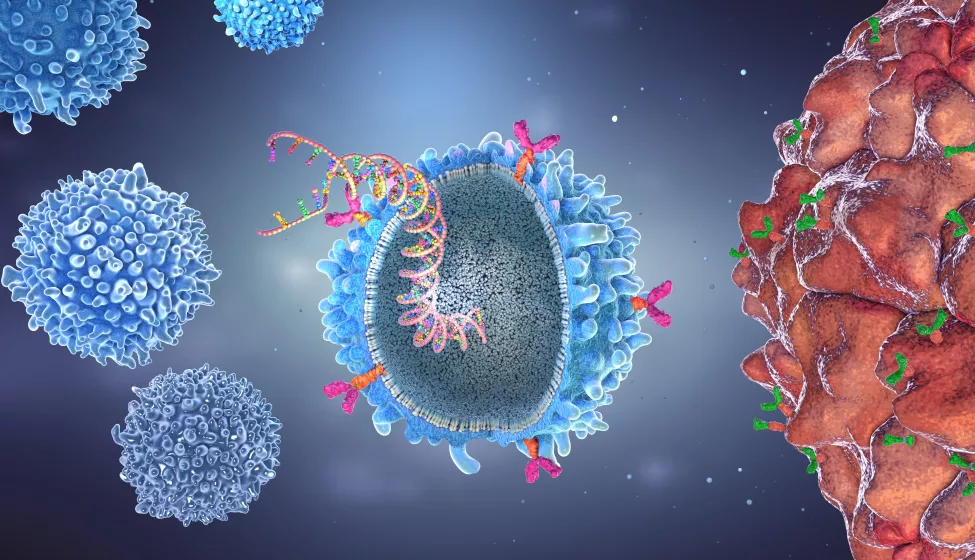
Biomanufacturing applies the principles of process engineering and chemical design to living systems. Living systems such as human cells can be engineered or modified outside of the body to produce a therapeutic product. CAR T-cell therapy involves collecting and using a patient's own immune cells to treat their malignancy in a customized fashion.
T cells collected from the patient are genetically modified in a lab to produce proteins on their surface called chimeric antigen receptors, or CARs. After enough modified T cells have been biomanufactured, they're infused back into the patient. If all goes well, the CAR T cells in the body will bind to specific proteins, or antigens, on the surface of the targeted cancer cells and destroy them.
This therapy's genesis dates to the early 1960s with the discovery of the origin of T cells — white blood cells known as lymphocytes that are involved in the immune system response — and research showing that immune cells could kill cancer in mice. Subsequent discoveries over the next 40 years helped establish the benefits of CAR T-cell therapy in humans.
Continued advances in the 2000s have resulted in regulatory approval of CAR T-cell therapy for a range of blood cancers, including s-cell lymphoma, acute lymphoblastic leukemia, and multiple myeloma. Clinical trials are underway to study CAR T-cell therapy for solid tumors, such as glioblastoma, colorectal, pancreatic, renal, hepatic, prostate, ovarian, breast, and thoracic cancer.

Companion diagnostics: safe and effective use of immunotherapy
In the late 1990s, during the development of Herceptin, an early immunotherapy drug, scientists recognized that only women whose breast tumors overexpressed the HER2 receptor were benefiting from the drug. Detecting how much HER2 was present and determining how much was necessary for response to the therapy required a new in vitro diagnostic (IVD) product to quantify HER2 accurately. In 1998, Herceptin and its CDx, HercepTest, received regulatory approval concurrently.
As precision medicine evolves in oncology, CDx are increasingly being adopted to guide treatment decisions. They can help provide critical patient information, such as identifying those most likely, or unlikely, to benefit from a specific CAR T-cell therapy and those likely to be at increased risk for serious side effects. Co-development and simultaneous approval of CDx-guided immunotherapy, including CAR T-cell therapy, is now standard practice in oncology.
But the co-development process still presents challenges. For example, when is the optimal time in therapeutic development to begin co-developing the CDx? Starting too early runs the risk that diagnostic development may outpace that of the therapy, and if the therapy eventually proves ineffective, the funding and resources devoted to the diagnostic may be of little or no value. In contrast, starting too late risks having an effective therapy ready to be submitted for approval before a CDx is ready for approval, thereby delaying market entry.
Another option is to use a CDx that is already approved and on the market. This can speed regulatory approval; in fact, it may be difficult to convince a regulatory body to use a non-approved newly developed diagnostic test rather than one that is appropriate and already on the market. However, this arrangement can present commercial challenges if the therapy is developed and marketed by one company and the CDx is developed and marketed by another. In addition, the existing diagnostic test may require a change in labeling for the new intended use.
Global regulatory agencies have published guidance around CDx, including the European Medical Association (Guidance on the procedural aspects of companion diagnostics), the U.S. Food and Drug Administration (Companion Diagnostics), and Germany's Federal Institute for Drugs and Medical Devices (Companion diagnostics).
Research and discovery are just the first steps in the complex path from development to regulatory approval and successful commercialization for CAR T-cell therapies and CDx
A complex path to market and beyond for therapies with companion diagnostics
The path to market for a CAR T-cell therapy and CDx is complex and challenging worldwide. Even after regulatory approval and launch, more challenges await. Here are a few examples:
- Regulatory requirements are still evolving, with new ones continually being added. The In Vitro Diagnostics Regulation (IVDR), which took effect in May 2022, increased the regulatory requirements in the European Union to assess CDx, which affects everyone in the development and delivery chain, including manufacturers, importers, and distributors. In October 2023, FDA proposed consolidating regulation of all IVDs in the U.S., including CDx. To date, there have been two IVD regulatory methods: manufacturer distributed IVDs, which FDA regulated directly, and laboratory developed tests (LDTs), which it has regulated indirectly through a general enforcement discretion approach. LDTs are tests that are designed, manufactured, and used within a single clinical laboratory that is certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA) for high-complexity testing.
- A recent examination found differences in the clinical validation requirements for CDx in Europe, Korea, and the U.S. Each regulatory body — EMA, MFDS, and FDA, respectively — has a common goal of ensuring accuracy and reliability based on the intended use of a CDx. However, each has unique requirements to achieve the goal. As one example, among several, FDA emphasizes the relevance of outcome values to the clinical reference standard, while the MFDS applies a risk-factor-based approach.
- Using biomanufacturing processes for personalized medicine such as CAR T-cell therapy pose a number of potential risks to both patients and workers that must be carefully controlled. For example, engineers and technicians on site developing and manufacturing these products may be exposed to hazardous chemical or biological substances, including bloodborne pathogens. Manufacturers must consider appropriate precautions, training, and education to protect their workers. The processes typically require highly trained personnel and rigorously maintained procedures to ensure the consistent quality of therapeutic products. Assessing risk to patients requires considering individual pathology when evaluating the therapeutic product's pharmacokinetic and pharmacodynamic behavior.
- Even when CAR T-cell therapies and CDx complete clinical trials and receive regulatory approval, there are barriers to widespread use. These include complex logistics, biomanufacturing infrastructure constraints, process engineering scale-up limitations, batch variability across manufacturing sites, toxicity concerns, and financial burdens. In addition, FDA is investigating the risk of T-cell malignancy secondary to CAR T-cell immunotherapies.
- The evidence of the long-term safety and efficacy of these therapies remains limited. As such, regulators often seek additional long-term safety and effectiveness data after regulatory approval. This requires manufacturers to consider study designs that enable long-term real-world data collection, including decentralized data collection to minimize the burden on patients and providers.
Because industry standards for CAR T-cell therapy and CDx are still developing, biopharmaceutical companies often do not have a benchmark or reference point to pursue during discovery and development. Consequently, a manufacturer must demonstrate safety and efficacy/effectiveness of a therapy and diagnostic both pre-approval and post-market with limited guidance through standardized or tried-and-tested methods.
This is particularly challenging, as it is the manufacturer's responsibility to verify and validate test methods, acceptance criteria, experimental design, biomanufacturing techniques, and other elements that can typically be found in industrial standards to control risks. Furthermore, because the available industry standards are either still being developed or are relatively new to the public, new evidence or unexpected complications could come to light. This may modify the existing process, thereby making even seemingly established standards a moving target.
Multiple regulatory bodies can increase complexity and time to market
In the U.S., IVD products, including CDx, are regulated by the Center for Devices and Radiological Health (CDRH) at the FDA. They are regulated in the same manner as other medical devices and subject to premarket and post-market controls. Once approved, an IVD product can be manufactured and distributed in the same manner as a medical device. Because CAR T-cell therapies are regulated by the Center for Biologics Evaluation and Research (CBER) at FDA, with only a handful of approvals on the market, the approval process for the combination product of the CAR T-cell therapy and CDx must include representatives from both centers. This can create difficulties for manufacturers who need to understand the processes and requirements for both centers.
LDTs currently represent an alternative path to regulatory approval for CDx in the U.S. An LDT is manufactured by and used within a single laboratory that is certified as CLIA-compliant under Centers for Medicare & Medicaid Services authority. There are pros and cons to selecting the regulatory approval process for manufacturer-distributed IVD products or LDTs, including differences in analytical and clinical validation, clinical utility, and commercial considerations. However, as previously noted, the choice may soon be irrelevant, since FDA has proposed a rule to explicitly regulate all IVDs, including LDTs, under the same framework as medical devices.
While approval in one country may be helpful in achieving regulatory approval in another, it doesn't guarantee it. For example, the EU has a similar approval process technically compared to the U.S., but it requires engagement with two regulatory entities: the European Medicines Agency (EMA) for therapeutics and a notified body for the diagnostic test (which also requires EMA input). There can also be additional regulatory requirements imposed by individual EU countries, and this is particularly important when considering factors such as obtaining patient samples from some Member States and testing them in others. Although the U.K. is currently aligned with past and present EU regulatory requirements (in some cases, making CDx regulation less stringent), future divergence from the EU cannot be ruled out. As previously noted, countries outside Europe and the U.S., such as Korea, may have different clinical validation requirements for CDx. These differences are important to consider in the development of a global regulatory approval strategy to market a CAR T-cell therapy and CDX.
The value of end-to-end expertise to address the complexity of co-marketed CAR T-cell therapies and CDx
Medical researchers are working at the cutting edge of cancer treatment, discovering potential cures that were little more than aspirations just a decade ago. However, research and discovery are just the first steps in the complex path from development to regulatory approval and successful commercialization for CAR T-cell therapies and CDx. Some of these products can be eligible for expedited regulatory pathways (e.g., breakthrough designation in the U.S.), but this still means that a company trying to bring its product to market as quickly as possible must ensure that R&D, manufacturing, quality, compliance, and the other required elements of a regulatory submission are ready for launch.
Because CAR T-cell therapy and CDx are relatively new in cancer treatment, ongoing real-world data collection and analysis is imperative to better understand long-term effectiveness and safety. To date, over a decade of patient data demonstrates that CAR T cells can induce prolonged remissions in patients with certain blood cancers, often with minimal long-term adverse events. Long-term data collection and analysis of patient outcomes from CAR T-cell therapy and CDx for blood cancers now and solid tumors in the future will help optimize therapeutic outcomes. The use of new approaches, such as using digital health technologies in decentralized clinical trials or studies, will make it less burdensome for patients and their providers to capture and derive valuable insights from real-world data.
Experts with multidisciplinary knowledge, expertise, and experience can help integrate digital technologies into clinical trials, prepare and execute a regulatory strategy for global agencies, scale biomanufacturing, and conduct long-term observational studies. Partnering with them can help speed the journey to market more efficiently and cost-effectively and enable better, more successful outcomes for patients.
What Can We Help You Solve?
Exponent's multidisciplinary team of biomedical engineers, health scientists, and regulatory experts can help guide CAR T-cell therapies and CDx through discovery and development to biomanufacturing and commercialization. We support a wide array of activities, including assay design, integration of digital health technologies into clinical trials, regulatory submissions, and long-term observational studies.

Biomedical Expertise for Therapeutic Areas
Multidisciplinary insights for a range of therapeutic product development challenges.

Regulatory Compliance for Medical Products
Experienced regulatory support for medical devices, pharmaceuticals, and combination products.

Biomedical Engineering Laboratories
Expert biomedical laboratory services, delivering evidence-based answers for your most complex challenges.

Biocompatibility & Biological Risk Assessment
Address biocompatibility challenges throughout the medical-device product lifecycle.
Biomedical Engineering & Sciences
Evaluate the performance of biologics, biomaterials, medical devices, and combination products.

Clinical & Pharmacoepidemiology
Rigorous pharmacoepidemiology studies and clinical trials.